
The researchers predict that metallic hydrogen might be a room-temperature superconductor and could possibly exist, once created, at normal pressures. “It’s not too surprising that it happens – I mean, it’s been predicted by theory for 80 years – but it’s nice that they’re finally seen it and now they can start getting more quantitative data,” says David Ceperley at the University of Illinois at Urbana-Champaign. As the compression intensified, it began to turn opaque and black.īut at 495 Gpa, the hydrogen was shiny and reflective, indicating its transformation into a metal (although the researchers can’t be sure whether it was solid or liquid). To prevent the diamonds from cracking, the tips were carefully polished to remove surface defects, heated to remove any residual internal stresses, and coated in alumina, an extremely hard compound of aluminium and oxygen that hydrogen can’t seep through.Īt relatively low pressures, the compressed solid hydrogen was transparent. They were able to obtain such a high pressure by crushing the solid hydrogen between the flattened tips of two synthetic diamonds. So, in fact, it would be a bit of a spectacular material, if we had it at ambient pressure.When Silvera and Dias managed to turn hydrogen metallic, it was at a pressure of 495 GPa, well beyond the 360 GPa of Earth’s core. – And then it still has exotic properties like the mobility of the proton. It’s a bit like the dream of materials technology. – It is a superconductor at room temperature, so there is no system that conducts electricity without resistance at room temperature. There is no other chemical system that stores such energy. – It stores a phenomenal chemical energy. This metallic hydrogen is a bit of an ultimate material. 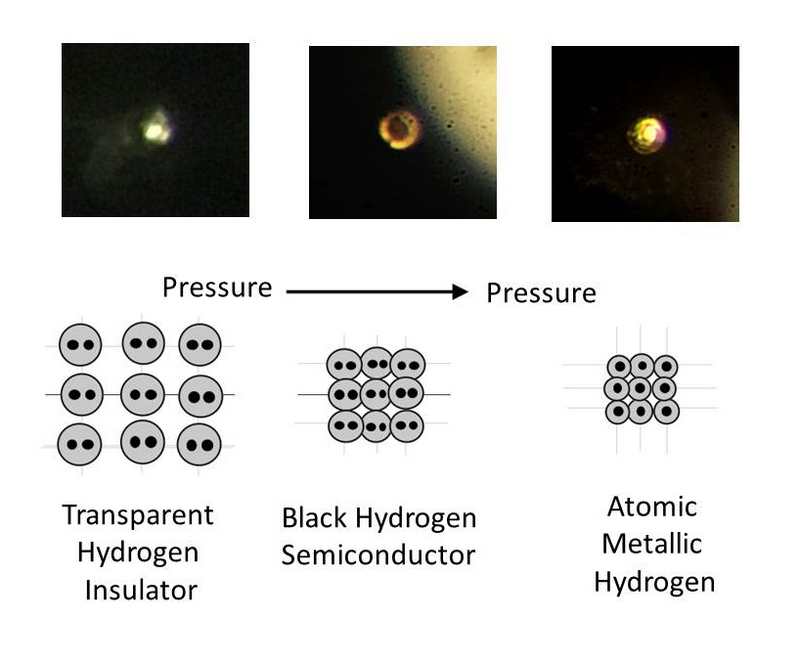
“There is no theory that can precisely describe the properties of hydrogen. 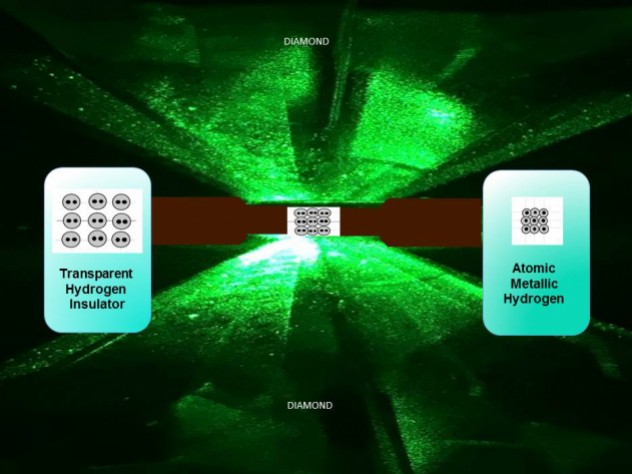
On a practical perspective, metallic hydrogen might achieve supra-conductivity: The study of this metal could learn a lot about the physics theory. Further research is needed for its “atomic” form. The sample obtained by the 3 French researchers was a cube of less than 5 µm on a side and was compressed between 2 diamond points. The result of 85 years of research! Properties of metallic Hydrogen It was not until Januthat Paul Loubeyre, Florent Occelli and Paul Dumas reached 425 GPa thanks to an anvil press, thus producing the famous metallic hydrogen. Mao and Hemley succeeded in understanding hydrogen at 250 GPa in 1989, a team from Cornell University reached 342 GPa in 1998. More recent research has estimated it at 400 or 620 gigapascals (GPa). This pressure was first estimated between 25 to 2,000 gigapascals. It’s possible existence was discovered in 1935! Eugene Wigner demonstrated that at sufficiently high pressures, hydrogen becomes a solid metal. While liquid hydrogen was first obtained in 1898, metallic hydrogen took much longer to develop. On Earth, we managed to make it in a lab recently : 2020! What is metallic Hydrogen ? There are indeed some in the depths of gas giants, such as Jupiter and Saturn. But did you know that it exists in … solid form? We know the hydrogen atom in gaseous form, its natural state, or liquid, used in particular in the Appolo spaceships, which brought humans to the Moon. This article is a part of our dossier on hydrogen, innovation and ecology. It is the material that could store the most energy and be superconducting… at room temperature. It is produced at pressures above 425 gigapascals. Metallic hydrogen was first “created” in 2020.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
